One of the most common chemical analyses performed on concrete is chloride analysis. Grouts, mortars, soils, and water samples are also often tested for chloride. Chloride is a negatively charged ion that has the ability to react with the iron in steel reinforcing, causing corrosion. This can lead to cracking of concrete due to the expansive nature of the corrosion, as well as failures of
structural steel due to loss of the steel integrity.

Chloride in concrete can come from many sources. Road deicing salts are a common source, and are usually a mixture of compounds including calcium chloride, sodium chloride, and magnesium chloride. Soil, adjacent to concrete, can also provide a source of chloride. Water used for mixing concrete can sometimes contain excessive amounts of chloride. As well, sometimes chloride is admixed into a concrete batch to speed up curing. Chloride in concrete is not necessarily a bad thing. But when there is steel reinforcement embedded in the concrete it can cause problems.

What does water-soluble chloride tell you? What about acid-soluble chloride? Water-soluble chloride represents the chloride that is available to cause corrosion right now. Acid-soluble chloride represents the chloride that is available to cause corrosion now, and chloride that is chemically bound and could potentially become available to cause corrosion.
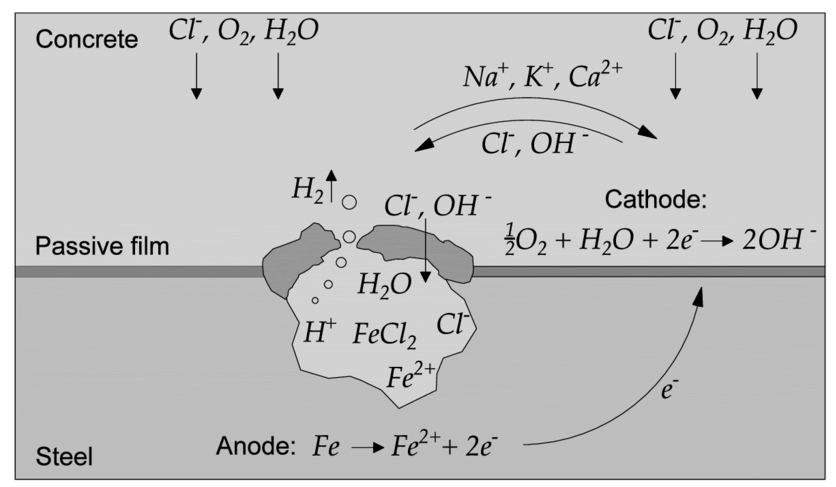
In a typical portland cement system, the high alkalinity of the cement forms a passivating oxide layer on the surface of the steel reinforcement, effectively protecting the steel reinforcing from moisture and oxygen-induced corrosion. Chloride ions present in the system can penetrate the passivating layer on the steel, and in high enough concentrations, promote corrosion if sufficient water and oxygen are also present. There are several different methods used for determining chloride content.
ASTM C1152 for determining acid-soluble chloride content
ASTM C1218 for determining water-soluble chloride content
ACI 222.1 for the soxhlet method, which determines water-soluble, chemically available chloride in aggregate.
ASTM C1556 for determining chloride diffusion coefficient of cementitious mixtures
The soxhlet method for determining chloride determines the chloride level of the aggregate, without crushing the aggregate. This method determines the water-soluble chloride that could become available to cause corrosion, and avoids over-estimation of chloride availability that can occur when analyzing crushed samples.
So what does each method tell you?
Water-soluble chloride analysis determines chloride that is readily available to promote corrosion. Acid-soluble chloride analysis determines the chloride that is readily available, plus the chloride that is chemically bound up in the paste. This is generally considered to be all the chloride that can possibly become available to cause corrosion, but there are situations where this will not be the case. Aggregate that contains chemically bound chloride is one of these situations. Performing acid-soluble chloride on aggregate containing chemically bound chloride can end up over-estimating the chemically available chloride because a sample is crushed to a fine powder prior to analysis, which allows chloride that would otherwise remain bound in the aggregate to be extracted.
The process of chloride analysis.
First let’s look at sampling for chloride analysis. For concrete, cores are taken in accordance with ASTM C42. In general, the diameter of the core should be three times the top nominal aggregate size for 1/4” profile slices to be analyzed. Smaller diameter cores can be used, but the concrete analyzed will have to be equivalent, so the thickness of the slice analyzed will have to be adjusted. For mortar and grout, a sample approximately the size of a golf ball is what we typically request. The nominal aggregate size in mortar is much smaller than concrete, so only a small sample is necessary to obtain a representative sample. When we receive and concrete core for chloride profile in the lab, we will cut the sample at the requested depths, dry, and crush the sample to a fine powder using the grinding mill. Once a sample is ground, it is then weigh and extracted using the requested method. The sample is filtered and the filtrate is analyzed for chloride. The titration used to determine chloride content is a potentiometric titration using a millivolt reader with a silver billet electrode and reference electrode to measure potential. Dilute silver nitrate solution is the titrant used to reach the equivalence point. The amount of silver nitrate used to reach the equivalence point is used to calculate the amount of chloride in the solution.
What do the results mean?
Knowing the chloride level in concrete or mortar can help determine several things.
-It can determine the potential for a mortar or concrete (or any other type of sample) to promote corrosion of reinforcing steel.
-It can help determine whether the concrete contains a chloride-bearing set accelerating admixture.
-It can help determine whether aggregates taken from marine sources were washed properly
Want a step-by-step guide on how to perform chloride analysis? Check out this article Step-by-Step Guide: Testing Chloride in Concrete with Silver Nitrate Titration
For more information contact Matthew Anderson at 847.306.9240 or manderson@materialsanalyticalgroup.com.


