Color measurement by colorimeter can be a powerful technique for analyzing different materials. A colorimeter measures the color of a material, and generates a numerical representation of the color in one of many available scales. Color can provide information such as whether a material is the color specified, or how different two colors are. As well, if certain criteria are met, color can be used to quantify materials in a mixture.

Differences in color are measured by differences in the numerical representation of the color. The advantage of using instrumental color measurement versus visual observation alone is that the subjectivity, in visual observation and interpretation of color, is removed.
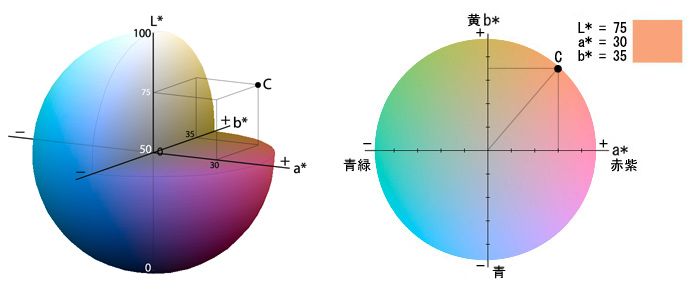
A commonly used color scale is the CIELAB system, first implemented in 1976, and is intended to approximate colors and color differences as observed visually. Color is measured with three coordinates: L* is the lightness, a* is the degree of redness or greenness, and b* is the degree of yellowness or blueness. The difference between two colors can be calculated according to ASTM D2244, Standard Practice for Calculation of Color Tolerances and Color Differences from Instrumentally Measured Color Coordinates. Changes in color can be measured by the difference in color (ΔE), using equation (1) below, or by the change in any of the individual coordinates. Literature on the CIELAB color system indicates that a ΔE of less than 1 – 3 is generally noticeable for most people (Billmeyer Jr. and Hammond III 1995) (Schanda 2007) .
ΔE = √[(L2 – L1)2 + (a2 – a1)2 + (b2-b1)2] (1)
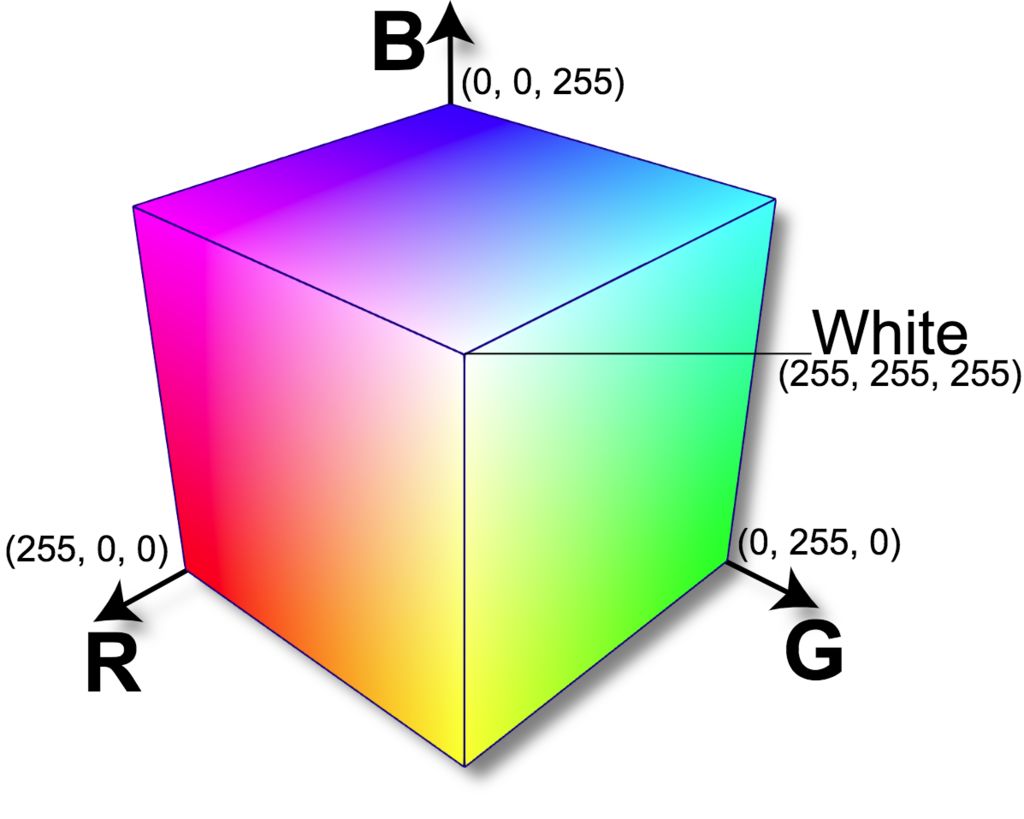
The RGB color scale is another common color system used. The R represents red, the G represents green, and the B represents blue. There is no variable that measures the intensity of light, or lightness in the RGB system. The RGB system is useful because there is a formula available for converting the RGB values measured by colorimeter into a single value, called the RGB Integer. This formula is shown is equation (2) below. Because the RGB Integer represents the color of a material as a single value, it can be useful for developing a calibration curve of concentration versus color.
RGB Integer = (R*65536) + (G*256) + B (2)

The RGB Integer was used for developing this method for determining bottom ash concentration in the coal power plant waste ponds, because it represents color as a single value. This value was directly compared to the concentration of the bottom ash in standards prepared to calculate the calibration curve. From the calibration curve prepared, a formula is derived that can convert color values to the concentration of bottom ash present in each sample analyzed.
In the field, the waste ponds were cleaned to depths determined by soil cores taken in and around the pond. After this, the waste ponds were divided up into nodes, and nodes were chosen randomly to check for levels of bottom ash. As well, any areas that looked particularly dark were sampled to test the bottom ash concentration. The threshold for the bottom ash at this location was determined to be 10% by mass. All EPA regulated contaminants would be below the EPA required limits at this threshold. To give the EPA and local Department of Environmental Quality (DEQ) more confidence that the contaminant levels would not exceed these required limits, a threshold of 5% bottom ash by mass of sample was set.
To prepare standards for developing a calibration curve of color versus concentration, bottom ash was sampled from stockpiles collected from the pond, and clean sand was sampled from the top of the pond where there was no bottom ash contamination. Both materials were examined microscopically to determine they were clean.

500 gram standards were prepared with bottom ash concentrations of 0%, 1%, 2%, 5%, and 10%. The standards were mixed and then color measured five times for each color measurement. Each measurement was the average of five color measurements. From 0% to 10%, the calibration curve was linear. From the prepared calibration curve, the color of a sample taken from the pond could be converted into the weight percent of bottom ash present in the sample. This concentration was confirmed microscopically using particle estimation charts for comparison. If there was a discrepancy between the color data and microscopic estimation, the microscopic estimation was deferred to, because the color was sometimes affected by local pockets of darker organic material. This was obvious when looking under the microscope, because there were no visible bottom ash particles.
At the conclusion of this project, the pond was cleaned out, the client was happy, and the DEQ was satisfied with the method and results. It was a rewarding method development project that helped our client, and the environment.
For more information contact Matt Anderson at 847.306.9240 or manderson@materialsanalyticalgroup.com.
References
Billmeyer Jr., Fred W., and Harry K. Hammond III. “Color and Light.” In Paint and Coating Testing Manual Fourteenth Edition of the Gardner-Sward Handbook, by Joseph V. Ed. Koeleske, 447-468. Philadelphia: ASTM International, 1995.
Schanda, Janos, Ed. Colorimetry Understanding the CIE System. Wiley Interscience, 2007.


