Unwanted water in buildings can cause staining, degradation of building materials, and an environment for mold to grow. Water can come from a few possible sources in a building, including drinking water, sewer plumbing, a leak starting at the exterior of the building envelope, ground water, and a pool if the building has one. The water chemistry will be different at each of these locations, which makes identifying the source of the leak possible. The location of the leak is also important in identifying the source.

To determine the water chemistry, ion chromatography is used to identify anions present in the water. Ion chromatography separates each ion by the ionic interactions of each analyte with an ion-exchange column. After separation, each ion concentration is determined by measuring the response of each analyte with a conductivity detector. Seven anions common to water (fluoride, chloride, nitrate, sulfate, nitrite, bromide, and phosphate) are analyzed, and the concentrations of each are determined. The water chemistry is determined for the leak water and each of the possible sources of the water.
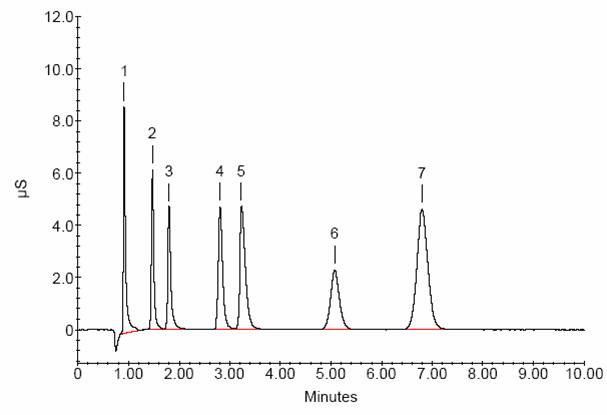
After determining the concentration of each ion, the source of the leak can be identified by the anion levels being consistent with one of the possible source waters. Sometimes leaks can come from more than one source, which will change the concentrations of the ions in solution. As well, leak water often migrates through and over concrete and other building materials, and can pick up ions along the way. These leak waters can be identified by comparing relative ratios of anions in the leak water versus the possible source waters.
For more information contact Matt Anderson at 847.306.9240 or manderson@materialsanalyticalgroup.com.


