Polyurethane foam is extensively and increasingly being used as insulation and vapor barriers in homes and buildings. There are many different types of polyurethane foam which makes it a versatile material, having a range of practical building applications. Types of foam include open cell polyurethane, closed cell polyurethane, extruded polystyrene, expanded polystyrene, spray polyurethane foam, and polyisocyanurate.

As a building material, the most commonly used polyurethanes are closed cell polyurethane foam, and spray polyurethane foam. Closed cell polyurethane is a high density rigid foam that typically provides an initial insulation R-value of R-5 per inch of foam. It is a vapor retarder with a typical perm rating of 1, which indicates it is a Class 3 vapor retarder, and is considered semi-permeable. Spray polyurethane foam is a a closed cell medium density rigid foam that is mixed and cured in the field. Typically the isocyanate and polyol that react to form polyurethane are mixed at the tip of a spray gun and sprayed into a wall cavity. The polyurethane then expands to fill the cavity before solidifying. Spray polyurethane foam can have an initial R-value of R-6.5 per inch, which decreases over time. Spray polyurethane foam has a perm rating of 1, indicating it is semi-permeable vapor retarder.

The range of applications of polyurethane have made it a popular building material, but polyurethane does have some inherent material characteristics that can be undesirable or even dangerous to the health of occupants of buildings insulated with polyurethane foam. These include expansion and contraction due to thermal changes (high CofTE), shrinkage over the lifetime of the material, susceptibility to cracking due to expansion stresses, off-gassing of solvents and precursor chemicals, the presence of chemical sensitizers, and an increased fire hazard.
Thermal Expansion and contraction of foam insulation
Coefficient of thermal expansion (CofTE) is the measure of a material’s expansion and contraction during heating a cooling cycles. Polyurethane foam used as an insulation material would be exposed to a wide range of temperatures, and thermal cycling would be expected often as the exterior of a building heats and cools throughout the day. According to literature, closed cell polyurethane foam can be expected to expand and contract up to 1/4 inch on a 96 inch board.. Therefore, if a building is insulated with polyurethane foam, air gaps could form that would cause drafts and loss of energy. This can be mitigated by using multiple layers of foam, with the seams offset and taped. Different polyurethane foams have different dimensional stability, so it is good to consult the literature for a specific product being used.
Shrinkage and cracking of spray polyurethane foam insulation
Spray polyurethane foam is mixed on site. As soon as the two parts that react to form polyurethane are mixed together, the curing process begins. The initial reaction makes a sticky, viscous fluid that sticks to almost anything, and can harden to a rigid materials in seconds. After the foam hardens, the curing process is not complete. The reaction will continue for hours, with unreacted precursors continuing to form polyurethane, and residual solvents evaporating. Manufacturers typically recommend allowing at least 24 hours before occupying a space that has had spray polyurethane foam installed. However, foam can cure at varying rates depending on the formulation, thickness of the application, temperature of the space, and the humidity of the space the foam is being installed in. Having a high initial temperature of the foam during installation and curing can also make the foam more susceptible to shrinking and cracking. This happens most often when there are two lifts of foams applied successively, causing a excess of heat from the curing process. In this situation, the foam would be hotter than typical, and harden while hot or expanded. Then when the foam cools down, there are extra stresses on the foam because the foam adhered to surfaces while it was hot or expanded. Foam is going to expand and contract with thermal changes, so the addition of more stress to the system significantly increases the chances of the foam cracking. Cracking significantly degrades the insulation values and the vapor barrier properties of foam.

chemicals used in the reaction to produce polyurethane foam
Polyurethane foams are formed by reacting an isocyanate (methylene diphenyl diisocyanate(MDI), toluene diisocyanate(TDI)) with an alcohol(diols, triols, polyols,) which would be identified as Part A and Part B by the manufacturer. The chemical structure and functional groups of the isocyanates and alcohols used in the preparation of the polyurethane determines the chemical and physical properties of the final product. Other additives are often added as reaction catalysts (amines), blowing agents, and flame retardants.
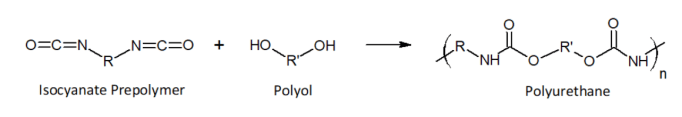
The isocyanate prepolymer used can be aromatic or aliphatic. Aliphatic isocynates are used for applications where exposure to UV light would be expected, such as coatings exposed to sunlight. Aromatic isocyanates are typically used in applications where discoloration of the polyurethane due to exposure to UV light is not important, such as insulation. Aromatic isocyanates are also cheaper which make them more prevalent. Aromatic isocyanates that are not used up in the reaction to produce polyurethane will be out-gassed over time as it migrates out of the foam. And if the foam was not mixed in the proper ratios of isocyanate and polyol then there can be high out-gassing of aromatic isocyanates. According to the EPA Methylene Diphenyl Diisocyanate (MDI) and Related Compounds Action Plan, isocyanate is a sensitizer chemical that is the leading cause of work-related asthma. A sensitizer is a chemical, that once sensitized, subsequent exposure to the chemical can trigger severe physiological reactions.
Catalysts used for the reaction that produces polyurethane are typically tertiary amines, such as triethylenediamine and dimethylcyclohexylamine. Amines can promote histamine liberation in people which can cause physiological symptoms similar to an allergic reaction. People exposed to amines have developed nausea, headaches, vomiting, rapid heart beat, and and weakness.
Flame retardant chemicals are prevalent in polyurethane foams because of the high flammability of the material. Since it is used as building insulation, it needs to meet the flammability requirements described in ASTM E84, Standard Test Method for Surface Burning Characteristics of Building Materials. Two of the common chemicals used as flame retardants are hexabromocyclododecane (HBCD), and Tris(1-chloro-2-propyl) phosphate (TCPP.) Both chemicals are considered persistant bioaccumulative toxins (PBTs), which are resistant to degradation, have high mobility, and are highly toxic. These chemicals are not chemically bound, and therefore can migrate out of the polyurethane foam over time.
Flammability of POlyurethane foam
Polyurethane foam is a flammable material, which needs flame retardant chemicals added to the reaction mixture in order to meet flammability requirements for building materials. Even with these chemicals added, polyurethane foam can still exacerbate fires. The video below from the Cellulose Insulation Manufacturer’s Association shows how quickly a polyurethane foam fire can spread. As well, as the foam burns, it will release the hazardous chemicals mentioned above, including the chemicals used as a flame retardant into the atmosphere.
Conclusion
Polyurethane foam is being increasingly used in modern construction, mainly because of it’s ease of use. There are potential issues of using polyurethane foam including the degrading R-value over time, the potential for shrinking and cracking, the fire hazard, and the potential out-gassing of hazardous chemicals. Improper installation makes many of these issues more likely to occur.
For more information contact Matt Anderson at 847.306.9240 or manderson@materialsanalyticalgroup.com.


